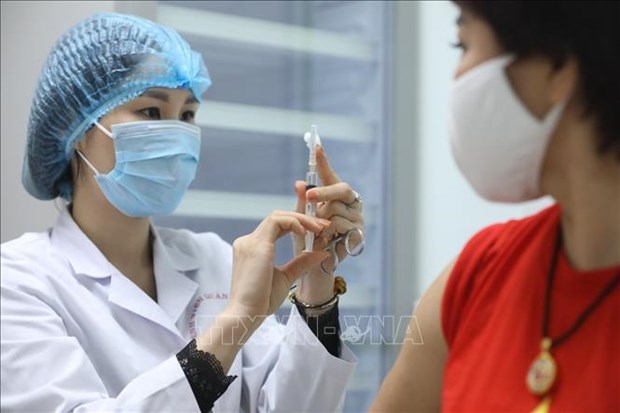
 A volunteer gets her 2nd shot of Nano Covax vaccine in the second trial phase (Photo: VNA)
A volunteer gets her 2nd shot of Nano Covax vaccine in the second trial phase (Photo: VNA)Hanoi (VNA)
– The Military Medical University under the Ministry of National Defence on March
25 began giving the second shots of the Nano Covax vaccine in the second trial phase to 26 volunteers who
receive the first jabs between February 26 and March 10.
Nano Covax developed by the Nanogen
Pharmaceutical Biotechnology Company is Vietnam’s first candidate vaccine to reach
the human trial stage.
According to Associate Prof. Dr. Chu Van Men, Director of the Military Medical University’s
Centre for Clinical Trials and Bioequivalence, after receiving the first shots,
exhibited symptoms such as pain at the injection point, a light fever, muscle aches, joint pain, and fatigue, but did not
require .
The health of the 560 volunteers remains stable, he said, adding that Nano
Covax is safe and the volunteers are ready for their second shots.
The second trial is being organised simultaneously at the Military Medical University
and the Pasteur Institute in Ho Chi Minh City, with volunteers including those
with mild background ailments such as hypertension, diabetes, and cardiovascular
issues.
As planned, the Military
Medical University will submit the preliminary test results to the Ministry of Health
and the National Council for Ethics in Biomedical Research in May for
review and evaluation, before conducting the third phase of trials.
Previously, at a
meeting of standing members of the National Steering Committee for COVID-19 Prevention
and Control, a representative from the Ministry of Health said that the third phase
of trials will take place from May to September, with the vaccine then registered
for circulation in September, three months earlier than scheduled.
Vietnam expects to have its locally-produced vaccine available by the end of the
third quarter./.
Source: VietnamPlus
